s75 10 300 increase (GE Healthcare)
Structured Review
S75 10 300 Increase, supplied by GE Healthcare, used in various techniques. Bioz Stars score: 94/100, based on 371 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more
https://www.bioz.com/result/s75 10 300 increase/product/GE Healthcare
Average 94 stars, based on 371 article reviews
Images
1) Product Images from "Design of amyloidogenic peptide traps"
Article Title: Design of amyloidogenic peptide traps
Journal: bioRxiv
doi: 10.1101/2023.01.13.523785

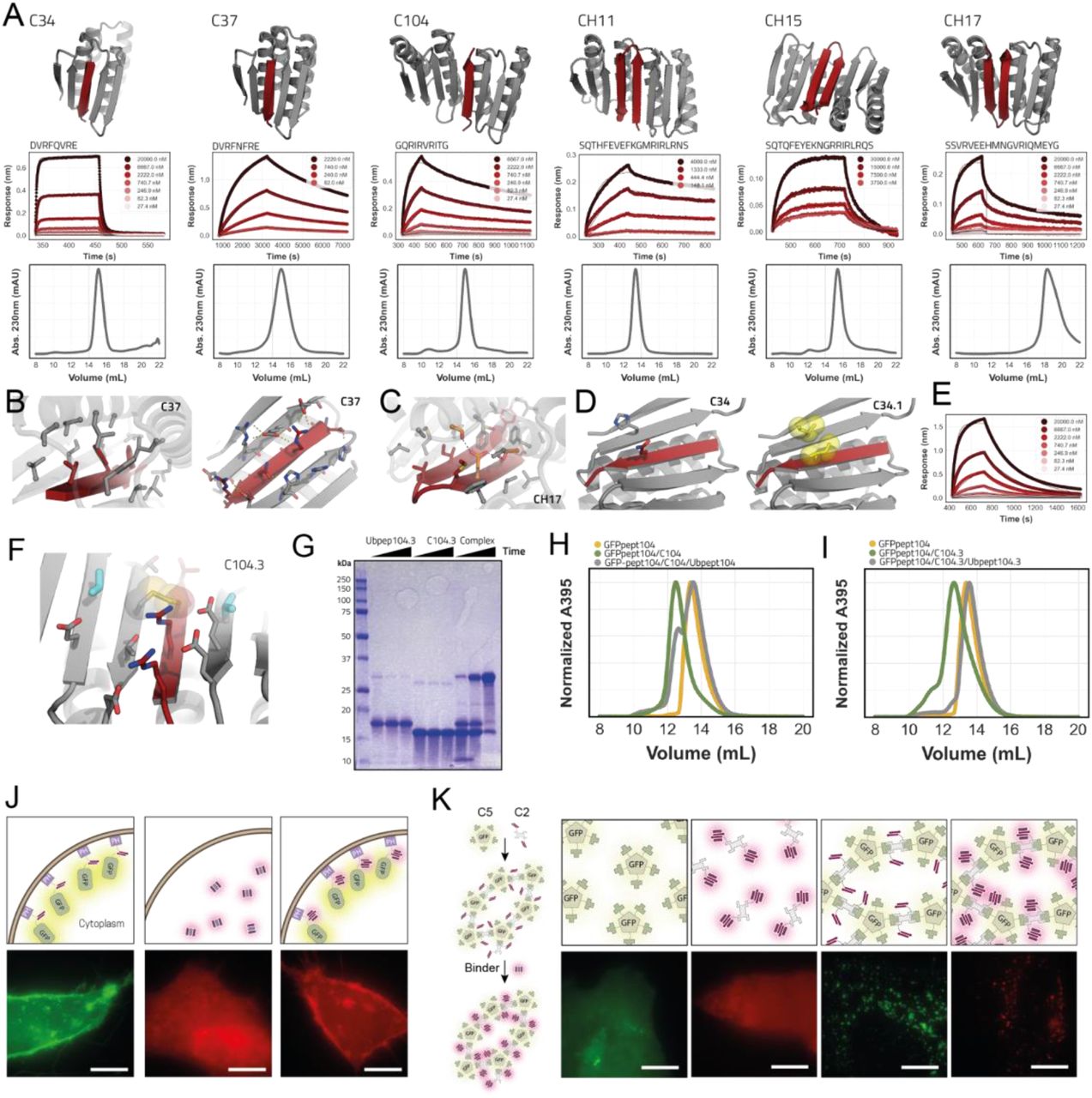
Figure Legend Snippet: a: Designed models for peptide binders (binder in gray, peptide in dark red). Respective BLI traces with kinetic fits and SEC (S75 increase 10/300) chromatograms of the binders are shown below the models. b: Detailed views of the solvent exposed interface of C37 (right) and the buried interface (left). C-alpha atoms as spheres. c: Detailed view of the buried part of the interface of hairpin binder CH17 with the designed hydrogen bond network depicted in orange sticks. d: Models of parent design C34 (left) and C34.1 (right) where an hydrophobic interaction pair (yellow sticks/spheres) is introduced to improve affinity. e: BLI trace of C34.1 binding its peptide that is immobilized on the biosensors. f: View of the designed interface disulfide on C104.3 (disulfide in spheres and sticks; additional redesigned residues in cyan). g: Non reducing SDS-PAGE gel showing disulfide formation (timepoints; t=0, t=90min t=overnight). h: SEC trace of preformed non-covalent C104 complex + GFP-pep104. i: SEC trace of preformed covalent disulfide linked C104.3 complex + GFP-pep104. j: Fluorescent microscopy images of mScartlet CH15.1 localization to membranes in HeLa cells. Scale bars 10 μm k: Fluorescent microscopy images of mScartlet CH15.1 localizing to designed intracellular GFP positive protein punctae in HeLa cells. Scale bars 10 μm.
Techniques Used: Binding Assay, SDS Page, Microscopy

Figure Legend Snippet: a: SEC binding assay showing that a fusion protein between GFP and 104 peptide binds to the C104 design on a S75 increase 10/300. b: Close-up view of the buried part of the C104 interface with Val6 shown in cyan sticks and spheres. Binder in gray and peptide in dark red. c: Biolayer interferometry trace of C104 binding to base peptide 104 and to a peptide with a V6R substitution. d: Interface close up view of C104 highlighting the hydrophobic-hydrophilic pattern of the peptide. Buried residues single letter amino acid identifiers are underlined.
Techniques Used: Binding Assay
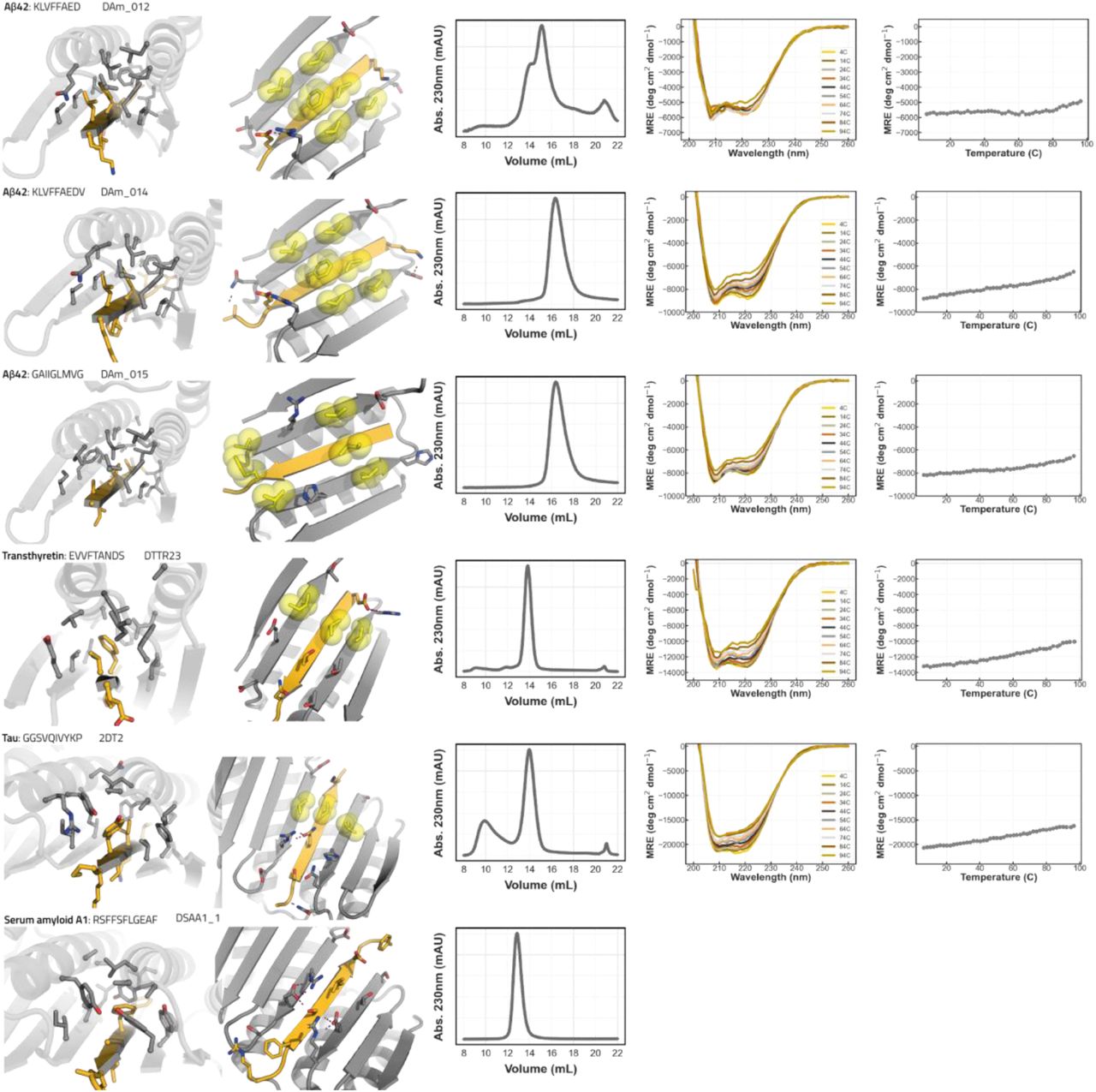
Figure Legend Snippet: Close-up view of solvent inaccessible part interface (first column), close up view of solvent accessible part of interface with hydrophobic interaction pairs in yellow spheres and sticks (2nd column), SEC trace of binder on S75 increase 10/300GL (3rd column), CD wavelength scans (4th column) and CD temperature melt at 222 nm. CD wavelength scans for DAm14 and DAm15 are the same as in main .
Techniques Used: